Endometritis & metritis
Discription of the disease
Endometritis and metritis are both inflammation of the uterus. Endometritis only involves the endometrium and the underlaying glandular tissues. Metritis involves the endometrium (lining of the uterus), the underlaying glandular tissues and the muscular layers. Metritis is often the word used for both types of inflammations, the difference between these two is mainly used theoretical. Since in practice it is difficult to distinguish.
Other disorders of the uterus which are closely related to endometritis and metritis are pyometra and lochiometra. An inflammation of the uterus due to a retained placenta, is called lochiometra. Pyometra is an uterus filled with pus, the cow does not come in heat because her endometrium is already filled with something (the pus).
Endometritis or ‘whites’ and metritis are common inflammations and infections of the uterine endometrium. Usually whites (white discharges from the vulva) are only visible when the cow is in heat, then is when she get rid of it. It is reported to have an incidence of between 10/15 percent in dairy herds (however it is very variable from herd to herd). It is a farm-leveled problem which affects the reproduction tract, so fertility, negatively.
Most cases of these uterine inflammations are indicated during period from calving to times of involution. Involution is the process by which the uterus returns to normal/non-pregnant size, this varies from 26-56 days after calving, with an average process time of 42-47 days. In these stressful times cows are very sensitive to infections, so then hygiene management is extra important.
Most cows with purulent discharges immediately postpartum are able to get rid of it within a few weeks, especially when these cows return to estrus in early lactation, this helps to flow out the infections from the uterus.
Metritis occurs in different stages, (sub)acute and chronic. Acute metritis (lochiometra, endometritis puerperalis acuta (short post partum)) appears quickly and generally it affects the cow’s appetite and milk production. Usually, acute metritis subsides after 2 or 3 weeks of detection. However, acute metritis may develop in to a chronic condition. Endometritis occurs three weeks or more after calving and it should not be confused with the more severe condition of acute metritis which occurs immediately post-partum. Chronic metritis (pyometra) persists over a long period and generally, the cow is not really visibly ill of it, usually she has a good appetite.
Various factors play a role in the severity of the uterus inflammation, such as the infectious agents, the degree and duration of the infection, the (metabolic) health state and the resistance of the cow. The prognosis is determined by the length of time that inflammation persists and the degree the infection. The infectious agent(s) and the degree of infection can be defined by microbial culture and uterine biopsy.
The disease can either be clinical or subclinical. In the ‘visible’ clinical state, symptoms of illness can be found, such as discharges, the size and thickness of the uterus or a fever (mainly in acute metritis). The subclinical type of metritis is not detectable by rectal palpation and no vaginal discharge is evident.
However, sometimes examination with a speculum will reveal a purulent discharge, but this is rarely done in practice. By microscopic examination of an uterine biopsy, subclinical endometritis can be positively diagnosed. (1)
Other disorders of the uterus which are closely related to endometritis and metritis are pyometra and lochiometra. An inflammation of the uterus due to a retained placenta, is called lochiometra. Pyometra is an uterus filled with pus, the cow does not come in heat because her endometrium is already filled with something (the pus).
Endometritis or ‘whites’ and metritis are common inflammations and infections of the uterine endometrium. Usually whites (white discharges from the vulva) are only visible when the cow is in heat, then is when she get rid of it. It is reported to have an incidence of between 10/15 percent in dairy herds (however it is very variable from herd to herd). It is a farm-leveled problem which affects the reproduction tract, so fertility, negatively.
Most cases of these uterine inflammations are indicated during period from calving to times of involution. Involution is the process by which the uterus returns to normal/non-pregnant size, this varies from 26-56 days after calving, with an average process time of 42-47 days. In these stressful times cows are very sensitive to infections, so then hygiene management is extra important.
Most cows with purulent discharges immediately postpartum are able to get rid of it within a few weeks, especially when these cows return to estrus in early lactation, this helps to flow out the infections from the uterus.
Metritis occurs in different stages, (sub)acute and chronic. Acute metritis (lochiometra, endometritis puerperalis acuta (short post partum)) appears quickly and generally it affects the cow’s appetite and milk production. Usually, acute metritis subsides after 2 or 3 weeks of detection. However, acute metritis may develop in to a chronic condition. Endometritis occurs three weeks or more after calving and it should not be confused with the more severe condition of acute metritis which occurs immediately post-partum. Chronic metritis (pyometra) persists over a long period and generally, the cow is not really visibly ill of it, usually she has a good appetite.
Various factors play a role in the severity of the uterus inflammation, such as the infectious agents, the degree and duration of the infection, the (metabolic) health state and the resistance of the cow. The prognosis is determined by the length of time that inflammation persists and the degree the infection. The infectious agent(s) and the degree of infection can be defined by microbial culture and uterine biopsy.
The disease can either be clinical or subclinical. In the ‘visible’ clinical state, symptoms of illness can be found, such as discharges, the size and thickness of the uterus or a fever (mainly in acute metritis). The subclinical type of metritis is not detectable by rectal palpation and no vaginal discharge is evident.
However, sometimes examination with a speculum will reveal a purulent discharge, but this is rarely done in practice. By microscopic examination of an uterine biopsy, subclinical endometritis can be positively diagnosed. (1)
Cause
A large number of microorganisms have been implicated as causes of metritis and endometritis: bacteria, viruses, fungi and protozoa have been cultured from uteri when metritis has been present.
The vagina hosts numerous microorganisms, in contrast to the sterile uterus. Opportunistic pathogens from the normal vaginal flora or from the environment may invade the uterus from time to time. A healthy uterus is able to rid itself of these kind of infections.
Several defence mechanisms exist for preventing (opportunistic) uterus infection. First of all, the vulva and cervix provide a physical barrier to opportunist bacteria. Thereby, bacteria which belong in the vagina prevent settling of pathogens, a higher pH level during oestrus prevent bacterial growth and various immunological mechanisms also act to prevent infection of the uterus.
In the immediate postpartum period, a stressful- and hard time for the cow, the uterus will get contaminated with several organisms. Subsequently, in most animals the uterine environment is re-established within days or weeks after calving. (3)
The vagina hosts numerous microorganisms, in contrast to the sterile uterus. Opportunistic pathogens from the normal vaginal flora or from the environment may invade the uterus from time to time. A healthy uterus is able to rid itself of these kind of infections.
Several defence mechanisms exist for preventing (opportunistic) uterus infection. First of all, the vulva and cervix provide a physical barrier to opportunist bacteria. Thereby, bacteria which belong in the vagina prevent settling of pathogens, a higher pH level during oestrus prevent bacterial growth and various immunological mechanisms also act to prevent infection of the uterus.
In the immediate postpartum period, a stressful- and hard time for the cow, the uterus will get contaminated with several organisms. Subsequently, in most animals the uterine environment is re-established within days or weeks after calving. (3)
Pathogenesis
The normally sterile uterus is contaminated by environmental microorganisms during parturition or immediately postpartum, these mmicroorganisms enter the uterus through various routes. Most commonly (opportunistic) organisms, mainly bacteria and fungi, contaminate the uterus during calving or early postpartum followed by a developing of chronic or subacute endometritis. Particularly in high productive cows and cows went through a bad maintained dry period, the reproductive system is very susceptible at this time: obviously, these cows deal with more stress and probably have a lower resistance related to a negative energy balance. Ureaplasms, mycoplasmas and Hemophilus somnus are common inhabitants of the vagina which can cause metritis in certain circumstances.
The main bacteria involved in endometritis is Trueperella pyogenes (arcanobacterium pyogenes), however, numerous gram-negative anaerobes (such as the Escherichia coli, Fusobacterium necrophorum and prevotella species) may also be involved. The presence of this opportunist bacteria can delay return to service and cyclical activity, prevent fertilisation and cause early embryonic death by producing a hostile uterine environment. It is also reported that it increases incidence of ovarian cysts.
Gram-negative bacteria have a thin cell wall of peptidoglycan, with an extra membrane around it to make the bacteria pathogenic. Anaerobe means it can live with and without oxygen. Anaerobic Gram-negative bacilli often act as opportunistic pathogens. Despite their thinner peptidoglycan, gram-negative bacteria are more resistant than gram-positive due to their relatively impermeable lipid based bacterial outer membrane. They are the most common anaerobes involved in infection and include some of the most antibiotic-resistant species.
Systemic infections, infections originating from elsewhere in the body, may result in metritis or endometritis as well. When cows suffer from infections like IBR (infectious bovine rhinotracheitis), BVD (bovine viral diarrhea) or leptospirosis which may spreads then to the uterus.
Less common types of infections are semen-borne infections entered the reproductive tract during natural breeding with a bull (venereal). The two common veneral diseases are campylobacteriosis (vibriosis) and trichomoniasis. Transmission of the infection may be by an infected bull or carried by the bull from an infected female to a susceptible cow. These infections are particularly dangerous if cows are inseminated when not in heat. It is important to know for sure that the cow is in heat when she get inseminated, since the resistance of the uterus is much lower in the period outside the period of heat as compared to during estrus. Artificial insemination studs are, generally, tested to be free of these diseases. (1,2,3,4,28)
The main bacteria involved in endometritis is Trueperella pyogenes (arcanobacterium pyogenes), however, numerous gram-negative anaerobes (such as the Escherichia coli, Fusobacterium necrophorum and prevotella species) may also be involved. The presence of this opportunist bacteria can delay return to service and cyclical activity, prevent fertilisation and cause early embryonic death by producing a hostile uterine environment. It is also reported that it increases incidence of ovarian cysts.
Gram-negative bacteria have a thin cell wall of peptidoglycan, with an extra membrane around it to make the bacteria pathogenic. Anaerobe means it can live with and without oxygen. Anaerobic Gram-negative bacilli often act as opportunistic pathogens. Despite their thinner peptidoglycan, gram-negative bacteria are more resistant than gram-positive due to their relatively impermeable lipid based bacterial outer membrane. They are the most common anaerobes involved in infection and include some of the most antibiotic-resistant species.
Systemic infections, infections originating from elsewhere in the body, may result in metritis or endometritis as well. When cows suffer from infections like IBR (infectious bovine rhinotracheitis), BVD (bovine viral diarrhea) or leptospirosis which may spreads then to the uterus.
Less common types of infections are semen-borne infections entered the reproductive tract during natural breeding with a bull (venereal). The two common veneral diseases are campylobacteriosis (vibriosis) and trichomoniasis. Transmission of the infection may be by an infected bull or carried by the bull from an infected female to a susceptible cow. These infections are particularly dangerous if cows are inseminated when not in heat. It is important to know for sure that the cow is in heat when she get inseminated, since the resistance of the uterus is much lower in the period outside the period of heat as compared to during estrus. Artificial insemination studs are, generally, tested to be free of these diseases. (1,2,3,4,28)
Diagnosis
Diagnose and treat metritis early in the post-calving period is very important to minimize the degree of inflammation and its effect/consequences. Diagnose every cow early in their lactation period, is good to do as part of a routine herd program. Thereby, diagnose the uterine inflammation in early stage minimizes long and costly dry periods.
An increase in size of the uterus wall may be detect (sub)clinical metritis as well by rectal palpation. The size of the uterus as related to time of calving; thickness (because it is an inflammation, the affected environment is red/swollen and thick) of the wall of the uterus and the presence, color, odor and consistency of fluid draining from one or both horns.
Other diagnostic techniques like examination on uterine culture or biopsy may be necessary, to see if there are any harmful bacteria/an inflammation for diagnosing clinical whether subclinical is present. Microscopic examination of the biopsy tissue can reveal the presence of acute or chronic metritis, this is especially valuable in case of future reproduction. Biopsy examination and uterine cultures both can positively confirm the presence of endometritis and the presence/absence of organisms in the uterus. This can avoid unnecessary intrauterine antibiotic treatment, which may have a detrimental effect on future reproduction and unnecessary treatment has little value when cultures are negative. In practice, these biopsies and microscopic examinations are not implemented.
An increase in size of the uterus wall may be detect (sub)clinical metritis as well by rectal palpation. The size of the uterus as related to time of calving; thickness (because it is an inflammation, the affected environment is red/swollen and thick) of the wall of the uterus and the presence, color, odor and consistency of fluid draining from one or both horns.
Other diagnostic techniques like examination on uterine culture or biopsy may be necessary, to see if there are any harmful bacteria/an inflammation for diagnosing clinical whether subclinical is present. Microscopic examination of the biopsy tissue can reveal the presence of acute or chronic metritis, this is especially valuable in case of future reproduction. Biopsy examination and uterine cultures both can positively confirm the presence of endometritis and the presence/absence of organisms in the uterus. This can avoid unnecessary intrauterine antibiotic treatment, which may have a detrimental effect on future reproduction and unnecessary treatment has little value when cultures are negative. In practice, these biopsies and microscopic examinations are not implemented.
Symptoms of clinical metritis
Clinical metritis and endometritis can frequently be diagnosed by the presence of a purulent vaginal discharge, by a veterinarian on rectal palpation.
One of the symptoms of clinical endometritis is mucopurulent vaginal discharge, which should be evident on vaginal exam 21 days or more post-calving. This discharge varies from white to yellow, but can also be pink or red and blood coloured. Dependent on severity, the odour discharge differs from offensive to relatively odourless. On an individual or herd level there may be a history of subfertility.
In contrast to chronic metritis, an animal suffering from acute metritis the animal may have a temperature and be obviously ill and this can progress to septicaemia. The discharge may be copious and have a offensive odour. Additionally, the afterbirth may be retained, or break off leaving a piece in the womb.
The discharge should not be confused with lochial discharge or vaginitis. Rectal palpation should reveal a poorly-involuted, oedematous uterus.
Notes:
One of the symptoms of clinical endometritis is mucopurulent vaginal discharge, which should be evident on vaginal exam 21 days or more post-calving. This discharge varies from white to yellow, but can also be pink or red and blood coloured. Dependent on severity, the odour discharge differs from offensive to relatively odourless. On an individual or herd level there may be a history of subfertility.
In contrast to chronic metritis, an animal suffering from acute metritis the animal may have a temperature and be obviously ill and this can progress to septicaemia. The discharge may be copious and have a offensive odour. Additionally, the afterbirth may be retained, or break off leaving a piece in the womb.
The discharge should not be confused with lochial discharge or vaginitis. Rectal palpation should reveal a poorly-involuted, oedematous uterus.
Notes:
- Inflammations of the cervix and vagina produce abnormal discharges as well, so further examination is necessary unless fluid can be palpated in the uterus.
- Whitish discharges from the vulva within 12 to 24 hours after natural breeding as well as small amounts of pus-like fluids on the insemination pipette are not necessary signs of metritis.
Consequences
The main consequence of endometritis is poor fertility in the future, particularly if not treated quickly. In the dairy sector, reproduction is essential: without calve no milk production, without milk production no income. Therefore this disease has a major economic impact by increasing calving interval, services per conception and decreasing milk yield. Increasing culling rates may be another effect of these.
The reproductive system, mainly the uterus and vagina, are damaged by the (cause of) inflammation and there is an increased production of endometrial fluid (functional loss and fluid retention so does not become pregnant), so the cow’s fertility will be less. Thereby, the cow will have a prolonged calving interval and increased insemination times.
Some cows (mainly the cows with acute metritis, will have a loss of appetite, which have an effect on the body condition (metabolism) and can thereby cause other disorders like a negative energy balance. (5,8)
The reproductive system, mainly the uterus and vagina, are damaged by the (cause of) inflammation and there is an increased production of endometrial fluid (functional loss and fluid retention so does not become pregnant), so the cow’s fertility will be less. Thereby, the cow will have a prolonged calving interval and increased insemination times.
Some cows (mainly the cows with acute metritis, will have a loss of appetite, which have an effect on the body condition (metabolism) and can thereby cause other disorders like a negative energy balance. (5,8)
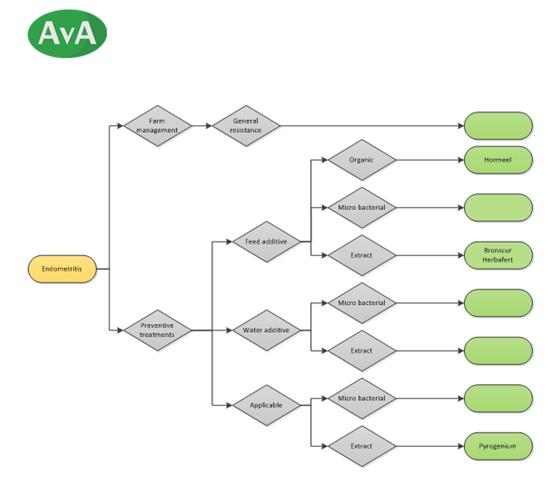
Decision tree
List of sources
1. Manspeaker, Dr. J.E., Metritis and endometritis IRM-22 - Dairy Integrated Reproductive Management, University of Maryland. Available from: <http://www.wvu.edu/~exten/infores/pubs/livepoul/dirm22.pdf> [24 May 2014]
2. Gilbert, R.O., Metritis and Endometritis in Large Animals, 2011. The Merck Veterinary Manual. Available from: < http://www.merckmanuals.com/vet/reproductive_system/metritis_in_large_animals/metritis_and_endometritis_in_large_animals.html#v3291743> [24 May 2014]
3. Endometritis – Cattle. Available from: <http://en.wikivet.net/Endometritis_-_Cattle> [24 May 2014]
4. Finegold, S.M. [1996], ‘Chapter 20 Anaerobic Gram-Negative Bacilli’ Medical Microbiology. 4th edition. Galveston (TX): University of Texas Medical Branch, Galveston
5. MSD Animal Health, Chronische baarmoederontsteking. Available from: http://www.rundvee-msd-animal-health.nl/vruchtbaarheid-en-reproductie-aandoening-baarmoederontsteking [24 May 2014]
6. Rodriguez-Martinez, H., Reproduction in Domestic Animals [2012] Reproduction in Domestic Animals. ISI Journal Citation Reports. Available from: < http://onlinelibrary.wiley.com/journal/10.1111/(ISSN)1439-0531 > [24 May 2014]
7. Manspeaker, Dr. J.E., Metritis and endometritis IRM-22 - Dairy Integrated Reproductive Management, University of Maryland. Available from: <http://www.wvu.edu/~exten/infores/pubs/livepoul/dirm22.pdf> [24 May 2014]
8. Gamroth, M. And Carroll, D. (1995) Dry Cow Feeding and Management. Available from: <
http://ir.library.oregonstate.edu/xmlui/bitstream/handle/1957/14790/em8624.pdf> [24 May 2014]
9. Cady, Dr. R.A., Dystocia—Difficult calving, what it costs and how to avoid it IRM-20 - Dairy Integrated Reproductive Management, University of New Hampshire. Available from: < http://www.wvu.edu/~exten/infores/pubs/livepoul/dirm20.pdf> [24 May 2014]
10. Causes of Dystocia. Integrated Livestock Management, Colorado State University. Available from: <http://www.cvmbs.colostate.edu/ilm/proinfo/calving/notes/causes.htm> [24 May 2014]
11. Mee, J.F. [April 2008], Prevalence and risk factors for dystocia in dairy cattle: A review. The Veterinary Journal Volume 176, Issue 1. Available from: < http://ac.els-cdn.com/S1090023307004285/1-s2.0-S1090023307004285-main.pdf?_tid=4363777e-e322-11e3-b226-00000aab0f02&acdnat=1400922385_49a66ca4a3e40baaafaa612ce719ed0c> pp. 93–101 [24 May 2014]
12. Cuneo, S.P., Card, C.S., Bicknell, E.J. [1993], Diseases of beef cattle associated with post-calving and breeding, Animal Care and Health Maintenance. Available from: http://ag.arizona.edu/arec/pubs/rmg/4%20animalcare&healthmaintenance/23%20diseasepostcalving93.pdf [24 May 2014]
13. Gilbert, R., van der Knaap, J., [2008] Gezond, maar onvruchtbaar. Veeteelt 1 September 2008. Available from: http://edepot.wur.nl/152414 [24 May 2014]
14. Nakao, T., [2001] Induction and synchronization of parturition in cattle, Arch. Tierz Dummerstorf 44 Special Issue. Laboratory of Animal Science, Graduale School for International Development and Cooperation, Hiroshima University, Japan. Available from: <http://arch-anim-breed.fbn-dummerstorf.de/pdf/2001/at01si1p145.pdf] pp. 145-150 [24 May 2014]
15. Opportunistic Pathogens. Available from: <http://www.slideshare.net/gurya87/opportunistic-pathogens> [24 May 2014]
16. Baarmoederontsteking. Zoetis. Available from: < https://www.zoetis.be/nl/conditions/rundvee/baarmoederontsteking.aspx> [24 May 2014]
17. Formularia antibiotica [2012], Werkgroep Veterinair Antibiotica Beleid – KNMVD. Available from: <http://wvab.knmvd.nl/wvab/formularia/formularia> [24 May 2014]
18. Juniperus sabina, Natural Medicinal Herbs. Available from: http://www.naturalmedicinalherbs.net/herbs/j/juniperus-sabina=savine.php [24 May 2014]
19. Vruchtbaarheidsproblemen, Virbac. Available from: <http://www.virbac.nl/producten/rund/algemene-gezondheidsinformatie/vruchtbaarheidsproblemen> [24 May 2014]
20. Groot M., Kleijer-Ligtenberg, G., van Asseldonk T., Hansma, H., [2011] Stalboekje Melkvee – Natuurlijk gezond met kruiden en andere natuurproducten. Available from: <http://edepot.wur.nl/162983> [24 May 2014]
21. Deru, J., Ellinger, L., Baars, T., [2006] Homeopathie bij rundvee een verkenning van behandelingen, succesfactoren en patroonherkenning in het kader van Bioveem – Bioveem Intern Rapport 16. Available from: http://www.centaurea.nl/wp-content/uploads/2010/04/Homeopathie-bij-rundvee.pdf [24 May 2014]
22. Baars, E., Baars, T., van Asseldonk, T., de Bruin, A., Ellinger, L. [2002] Deskstudie homeopathie en fytotherapie in de biologische veehouderij. Available from: <http://www.louisbolk.org/downloads/1302.pdf> [24 May 2014]
23. Homeopathie stalkaart bij koeien, Louis Bolk Instituut. Available at: <http://www.louisbolk.org/downloads/1903.pdf> [24 May 2014]
24. Hunter, F. (2004) Everyday homeopathy for animals, Beaconsfield: Beaconsfield Publishers LTD.
25. Meijer, I., [2014] Animal Health, lectures and extra help at van Hall Larenstein, Wageningen in April, May and June 2014.
26. Finke, E. (2011) Bedrijfsnetwerk op weg naar minder antibiotica. Available from: < http://edepot.wur.nl/185222> [24 May 2014]
27. Laven, R. (2014) Fertility in Dairy Herds – Uterine infection. Available from: http://www.nadis.org.uk/bulletins/fertility-in-dairy-herds/part-7-uterine-infection.aspx [24 May 2014]
28. Haynes, B.N. (2001) Keeping livestock healthy, Versa Press USA.
2. Gilbert, R.O., Metritis and Endometritis in Large Animals, 2011. The Merck Veterinary Manual. Available from: < http://www.merckmanuals.com/vet/reproductive_system/metritis_in_large_animals/metritis_and_endometritis_in_large_animals.html#v3291743> [24 May 2014]
3. Endometritis – Cattle. Available from: <http://en.wikivet.net/Endometritis_-_Cattle> [24 May 2014]
4. Finegold, S.M. [1996], ‘Chapter 20 Anaerobic Gram-Negative Bacilli’ Medical Microbiology. 4th edition. Galveston (TX): University of Texas Medical Branch, Galveston
5. MSD Animal Health, Chronische baarmoederontsteking. Available from: http://www.rundvee-msd-animal-health.nl/vruchtbaarheid-en-reproductie-aandoening-baarmoederontsteking [24 May 2014]
6. Rodriguez-Martinez, H., Reproduction in Domestic Animals [2012] Reproduction in Domestic Animals. ISI Journal Citation Reports. Available from: < http://onlinelibrary.wiley.com/journal/10.1111/(ISSN)1439-0531 > [24 May 2014]
7. Manspeaker, Dr. J.E., Metritis and endometritis IRM-22 - Dairy Integrated Reproductive Management, University of Maryland. Available from: <http://www.wvu.edu/~exten/infores/pubs/livepoul/dirm22.pdf> [24 May 2014]
8. Gamroth, M. And Carroll, D. (1995) Dry Cow Feeding and Management. Available from: <
http://ir.library.oregonstate.edu/xmlui/bitstream/handle/1957/14790/em8624.pdf> [24 May 2014]
9. Cady, Dr. R.A., Dystocia—Difficult calving, what it costs and how to avoid it IRM-20 - Dairy Integrated Reproductive Management, University of New Hampshire. Available from: < http://www.wvu.edu/~exten/infores/pubs/livepoul/dirm20.pdf> [24 May 2014]
10. Causes of Dystocia. Integrated Livestock Management, Colorado State University. Available from: <http://www.cvmbs.colostate.edu/ilm/proinfo/calving/notes/causes.htm> [24 May 2014]
11. Mee, J.F. [April 2008], Prevalence and risk factors for dystocia in dairy cattle: A review. The Veterinary Journal Volume 176, Issue 1. Available from: < http://ac.els-cdn.com/S1090023307004285/1-s2.0-S1090023307004285-main.pdf?_tid=4363777e-e322-11e3-b226-00000aab0f02&acdnat=1400922385_49a66ca4a3e40baaafaa612ce719ed0c> pp. 93–101 [24 May 2014]
12. Cuneo, S.P., Card, C.S., Bicknell, E.J. [1993], Diseases of beef cattle associated with post-calving and breeding, Animal Care and Health Maintenance. Available from: http://ag.arizona.edu/arec/pubs/rmg/4%20animalcare&healthmaintenance/23%20diseasepostcalving93.pdf [24 May 2014]
13. Gilbert, R., van der Knaap, J., [2008] Gezond, maar onvruchtbaar. Veeteelt 1 September 2008. Available from: http://edepot.wur.nl/152414 [24 May 2014]
14. Nakao, T., [2001] Induction and synchronization of parturition in cattle, Arch. Tierz Dummerstorf 44 Special Issue. Laboratory of Animal Science, Graduale School for International Development and Cooperation, Hiroshima University, Japan. Available from: <http://arch-anim-breed.fbn-dummerstorf.de/pdf/2001/at01si1p145.pdf] pp. 145-150 [24 May 2014]
15. Opportunistic Pathogens. Available from: <http://www.slideshare.net/gurya87/opportunistic-pathogens> [24 May 2014]
16. Baarmoederontsteking. Zoetis. Available from: < https://www.zoetis.be/nl/conditions/rundvee/baarmoederontsteking.aspx> [24 May 2014]
17. Formularia antibiotica [2012], Werkgroep Veterinair Antibiotica Beleid – KNMVD. Available from: <http://wvab.knmvd.nl/wvab/formularia/formularia> [24 May 2014]
18. Juniperus sabina, Natural Medicinal Herbs. Available from: http://www.naturalmedicinalherbs.net/herbs/j/juniperus-sabina=savine.php [24 May 2014]
19. Vruchtbaarheidsproblemen, Virbac. Available from: <http://www.virbac.nl/producten/rund/algemene-gezondheidsinformatie/vruchtbaarheidsproblemen> [24 May 2014]
20. Groot M., Kleijer-Ligtenberg, G., van Asseldonk T., Hansma, H., [2011] Stalboekje Melkvee – Natuurlijk gezond met kruiden en andere natuurproducten. Available from: <http://edepot.wur.nl/162983> [24 May 2014]
21. Deru, J., Ellinger, L., Baars, T., [2006] Homeopathie bij rundvee een verkenning van behandelingen, succesfactoren en patroonherkenning in het kader van Bioveem – Bioveem Intern Rapport 16. Available from: http://www.centaurea.nl/wp-content/uploads/2010/04/Homeopathie-bij-rundvee.pdf [24 May 2014]
22. Baars, E., Baars, T., van Asseldonk, T., de Bruin, A., Ellinger, L. [2002] Deskstudie homeopathie en fytotherapie in de biologische veehouderij. Available from: <http://www.louisbolk.org/downloads/1302.pdf> [24 May 2014]
23. Homeopathie stalkaart bij koeien, Louis Bolk Instituut. Available at: <http://www.louisbolk.org/downloads/1903.pdf> [24 May 2014]
24. Hunter, F. (2004) Everyday homeopathy for animals, Beaconsfield: Beaconsfield Publishers LTD.
25. Meijer, I., [2014] Animal Health, lectures and extra help at van Hall Larenstein, Wageningen in April, May and June 2014.
26. Finke, E. (2011) Bedrijfsnetwerk op weg naar minder antibiotica. Available from: < http://edepot.wur.nl/185222> [24 May 2014]
27. Laven, R. (2014) Fertility in Dairy Herds – Uterine infection. Available from: http://www.nadis.org.uk/bulletins/fertility-in-dairy-herds/part-7-uterine-infection.aspx [24 May 2014]
28. Haynes, B.N. (2001) Keeping livestock healthy, Versa Press USA.